AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Nitrogen xps peak3/31/2024  Therefore, the peak intensities give information about the percent composition of a material. For example, if a peak, A, is half the height of another peak B, that means there were half as many electrons detected with the binding energy at A compared to the number of electrons detected with the binding energy at B. The shorter the peak, the less electrons represented. Peaks from the XPS spectra give relative number of electrons with a specific binding energy. (Figure courtesy of the creative commons license. 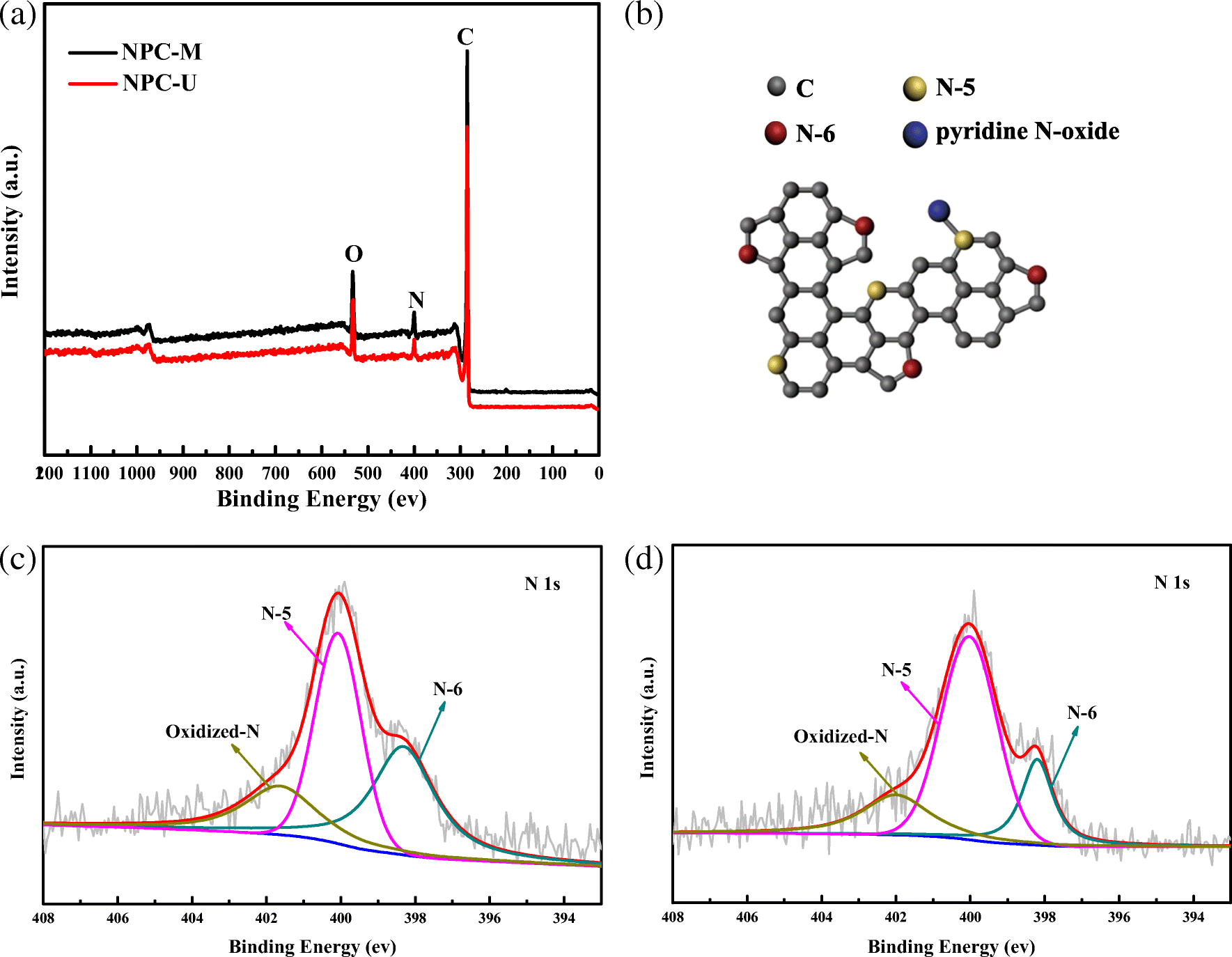
Graph showing the binding energies of electrons from different orbitals (F1s, O1s, Si2p, etc.) and their intensities which tell the atomic composition of the sample based on the amounts of each electron from different orbitals present. Electrons of different energies follow different paths through the detector which allows the computer to differentiate the electrons and produce the spectra seen below.įigure \(\PageIndex\): XPS spectra. 
The energy of those ejected electrons is analyzed by a detector and a plot of these energies and relative numbers of electrons is produced. X-rays (photons) are shot onto a sample, and when electrons in the sample absorb enough energy, they are ejected from the sample with a certain kinetic energy. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
